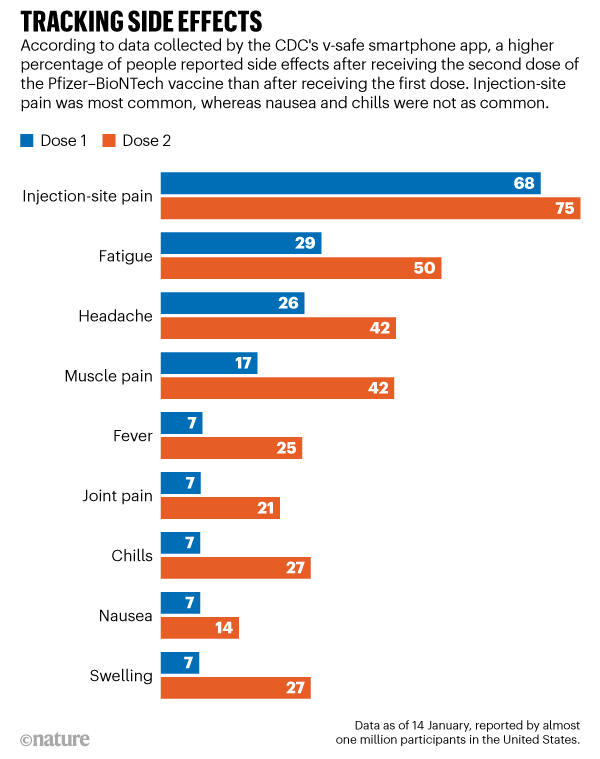
Very common (10% or more): Fatigue (up to 75.2%), pain (up to 71.4%), use of antipyretic/pain medication (up to 57.3%), axillary swelling (up to 28%), axillary tenderness (up to 28%), fever (up to 23.9%)Ĭommon (1% to 10%): Axillary/groin swelling or tenderness Psychiatric Postmarketing reports: Dizziness, facial paralysis, hypoesthesia, paresthesia OtherĪxillary swelling and tenderness included localized axillary swelling or tenderness ipsilateral to the vaccinated arm and lymphadenopathy. Very common (10% or more): Headache (up to 78.4%), sleepiness (up to 37.3%)įrequency not reported: Acute peripheral facial paralysis/palsy, Bell's palsy, febrile convulsion Very common (10% or more): Myalgia (up to 61.5%), chills (up to 45.4%), arthralgia (up to 41%) Nervous systemĪcute peripheral facial paralysis/palsy was reported in 3 patients who received their second dose after 22 days, 28 days, and 32 days. Very common (10% or more): Appetite loss (up to 32.1%) Musculoskeletal Very common (10% or more): Injection site pain (up to 97.2%), localized swelling/hardness (up to 17%), injection site erythema (up to 13.5%), localized swelling/induration (up to 12.6%)Ĭommon (1% to 10%): Delayed injection site reactions, injection site lymphadenopathyįrequency not reported: Injection site pruritus, injection site rash, injection site swelling, injection site urticaria Metabolic 
Localĭelayed injection site reactions included at least 1 of the following: erythema, pain, and/or swelling.įacial swelling occurred in 2 patients on Day 1 and Day 3 with a history of injection of dermatological fillers. Postmarketing reports: Anaphylaxis ImmunologicĬommon (1% to 10%): Acute otitis media Īcute otitis media occurred more frequently in patients 6 to 23 months of age who received this vaccine compared to placebo. Very common (10% or more): Nausea (up to 24%), vomiting (up to 24%) HematologicĬommon (1% to 10%): Lymphadenopathy HypersensitivityĬommon (1% to 10%): Hypersensitivity events/hypersensitivity Postmarketing reports: Acute urticaria, delayed urticaria, erythema multiforme Gastrointestinal Postmarketing reports: Myocarditis, pericarditis Dermatologic The most commonly reported side effects included injection site pain, headache, fatigue, and myalgia. Bleeding, blistering, burning, coldness, discoloration of the skin, feeling of pressure, hives, infection, inflammation, itching, lumps, numbness, pain, rash, redness, scarring, soreness, stinging, swelling, tenderness, tingling, ulceration, or warmth at the injection siteĪpplies to sars-cov-2 (covid-19) mrna-1273 vaccine: intramuscular suspension.Also, your health care professional may be able to tell you about ways to prevent or reduce some of these side effects.Ĭheck with your health care professional if any of the following side effects continue or are bothersome or if you have any questions about them: Incidence not known These side effects may go away during treatment as your body adjusts to the medicine. Some side effects of sars-cov-2 (covid-19) mrna-1273 vaccine may occur that usually do not need medical attention. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
